Search Thermo Fisher Scientific
Page Contents
- Phosphatase Substrates Yielding Soluble Fluorescent Products
- ELF 97 Phosphate: A Phosphatase Substrate That Yields a Fluorescent Precipitate
- BODIPY FL ATP-γ-S and BODIPY FL GTP-γ-S Thioesters: Substrates for the Fhit Nucleotide-Binding Protein
- Chemiluminescent Phosphatase Substrates
- Chromogenic Phosphatase Substrate
- Kits for Detecting Phosphatases, Polymerases and Nucleases
- Antibody Beacon Tyrosine Kinase Assay Kit
- ATP Determination Kit
- Data Table
- Ordering Information
Cells utilize a wide variety of phosphate and polyphosphate esters as enzyme substrates, second messengers, membrane structural components and vital energy reservoirs. This section includes an assortment of reagents and methods for detecting the metabolism of phosphate esters. Our diverse array of fluorogenic, chemiluminescent and chromogenic substrates include substrates for phosphatases, as well as reagents to measure the activity of enzymes such as ATPases, GTPases, DNA and RNA polymerases and paraoxonase. Unlike phosphatases, which hydrolyze phosphate esters, kinases use ATP to phosphorylate their target. Our Antibody Beacon Tyrosine Kinase Assay Kit (A35725, described below) provides a general and high-throughput solution assay for measuring the activity of tyrosine kinases using unlabeled peptides. In addition to these reagents, we offer several nucleotide analogs and substrates for phosphodiesterases and phospholipases that are described in Probes for Protein Kinases, Protein Phosphatases and Nucleotide-Binding Proteins—Section 17.3 and Probes for Lipid Metabolism and Signaling—Section 17.4, respectively.
By far the largest group of chromogenic and fluorogenic substrates for phosphate-ester metabolizing enzymes are those for simple phosphatases such as alkaline and acid phosphatase, both of which hydrolyze phosphate monoesters to an alcohol and inorganic phosphate. Conjugates of calf intestinal alkaline phosphatase are extensively used as secondary detection reagents in ELISAs,![]() immunohistochemical techniques
immunohistochemical techniques ![]() and Southern, northern and western blot analyses. In addition, phosphatases serve as enzyme markers, allowing researchers to identify primordial germ cells,
and Southern, northern and western blot analyses. In addition, phosphatases serve as enzyme markers, allowing researchers to identify primordial germ cells,![]() to distinguish subpopulations of bone marrow stromal cells
to distinguish subpopulations of bone marrow stromal cells ![]() and to investigate in vitro differentiation in carcinoma cell lines.
and to investigate in vitro differentiation in carcinoma cell lines.![]() P ALP-1, the gene for human placental alkaline phosphatase, has been used as a eukaryotic reporter gene that is superior to lacZ for lineage studies in murine retina.
P ALP-1, the gene for human placental alkaline phosphatase, has been used as a eukaryotic reporter gene that is superior to lacZ for lineage studies in murine retina.![]() This gene has also been engineered to produce a secreted alkaline phosphatase (SEAP), allowing quantitation of gene expression without disrupting the cells.
This gene has also been engineered to produce a secreted alkaline phosphatase (SEAP), allowing quantitation of gene expression without disrupting the cells.![]()
Fluorescein Diphosphate
First described in 1963,![]() fluorescein diphosphate (FDP, F2999) is perhaps the most sensitive fluorogenic phosphatase substrate available. The colorless and nonfluorescent FDP reagent is hydrolyzed to fluorescein (F1300, F36915; Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1), which exhibits superior spectral properties (EC ~90,000 cm-1M-1, quantum yield ~0.92). We have succeeded in preparing a highly purified FDP and find it to be an excellent substrate for alkaline phosphatase in ELISAs, providing detection limits at least 50 times lower than those obtained with the chromogenic 4-nitrophenyl phosphate.
fluorescein diphosphate (FDP, F2999) is perhaps the most sensitive fluorogenic phosphatase substrate available. The colorless and nonfluorescent FDP reagent is hydrolyzed to fluorescein (F1300, F36915; Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1), which exhibits superior spectral properties (EC ~90,000 cm-1M-1, quantum yield ~0.92). We have succeeded in preparing a highly purified FDP and find it to be an excellent substrate for alkaline phosphatase in ELISAs, providing detection limits at least 50 times lower than those obtained with the chromogenic 4-nitrophenyl phosphate.![]() The relatively high pH required to monitor alkaline phosphatase activity is advantageous because it also enhances fluorescein's fluorescence. FDP has been used for a diverse set of applications, including:
The relatively high pH required to monitor alkaline phosphatase activity is advantageous because it also enhances fluorescein's fluorescence. FDP has been used for a diverse set of applications, including:
- Assaying alkaline phosphatase in a microfluidic device
- Assaying diarrheic shellfish toxins in a microplate assay
- Detecting alkaline phosphatase immobilized on microspheres in an optical sensor array
- Measuring endogenous phosphatase in an assay for cell adhesion and migration that is reported to be as sensitive as 51Cr-release assays
- Quantitating the effect of inhibitors on tyrosine phosphatases
Dimethylacridinone (DDAO) Phosphate
Our 7-hydroxy-9H-(1,3-dichloro-9,9-dimethylacridin-2-one (DDAO phosphate, D6487) phosphatase substrate yields a hydrolysis product that is efficiently excited by the 633 nm spectral line of the He-Ne laser to produce bright red fluorescence with absorption/emission maxima of ~646/659 nm (Figure 10.3.1). Although the substrate itself is fluorescent, the difference between the substrate's excitation maximum and that of the phenolic hydrolysis product is over 200 nm (Figure 10.3.2), allowing the two species to be easily distinguished. Like other DDAO-based substrates, DDAO phosphate has good water solubility, a low KM and a high turnover rate, making it particularly useful for both fluorescence- and absorption-based microplate assays.![]()
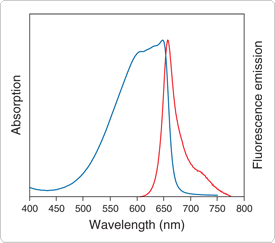
Figure 10.3.1 Normalized absorption and fluorescence emission spectra of DDAO, which is formed by alkaline phosphatase–mediated hydrolysis of DDAO phosphate (D6487).
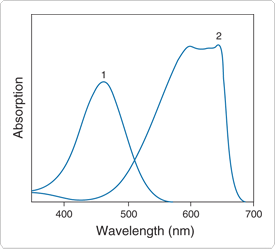
Figure 10.3.2 Absorption spectra of 1) DDAO galactoside (D6488) and 2) DDAO (H6482) at equal concentrations in pH 9 aqueous buffer. These spectra show the large spectral shift accompanying enzymatic cleavage of DDAO-based substrates. DDAO phosphate (D6487) has very similar spectra to DDAO galactoside.
Methylumbelliferyl Phosphate (MUP) and Difluorinated Methylumbelliferyl Phosphate (DiFMUP)
We offer 4-methylumbelliferyl phosphate (MUP, M6491), a widely used fluorogenic substrate for alkaline phosphatase detection as either a free acid. MUP has been used for a variety of ELISA protocols ![]() in which the relatively high pH optimum of alkaline phosphatase permits continuous detection of the rate of formation of 4-methylumbelliferone (7-hydroxy-4-methylcoumarin, H189, Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1). MUP has also been used to count cells based on their alkaline phosphatase activity,
in which the relatively high pH optimum of alkaline phosphatase permits continuous detection of the rate of formation of 4-methylumbelliferone (7-hydroxy-4-methylcoumarin, H189, Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1). MUP has also been used to count cells based on their alkaline phosphatase activity,![]() to detect PCR amplification products
to detect PCR amplification products ![]() and to identify and characterize bacteria.
and to identify and characterize bacteria.![]()
We also offer 6,8-difluoro-4-methylumbelliferyl phosphate (DiFMUP, D6567, D22065), which exhibits extraordinary spectral properties that are advantageous for the assay of both acid and alkaline phosphatase activity and is probably the best general substrate available for measuring the activity of the protein phosphatases that are important for high-throughput screening applications under physiological conditions.![]() The hydrolysis product of DiFMUP—6,8-difluoro-4-methylumbelliferone (6,8-difluoro-7-hydroxy-4-methylcoumarin, D6566; Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1)—exhibits both a lower pKa (4.9 versus 7.8, Figure 10.3.3) and a higher fluorescence quantum yield (0.89 versus 0.63) than the hydrolysis product of MUP.
The hydrolysis product of DiFMUP—6,8-difluoro-4-methylumbelliferone (6,8-difluoro-7-hydroxy-4-methylcoumarin, D6566; Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1)—exhibits both a lower pKa (4.9 versus 7.8, Figure 10.3.3) and a higher fluorescence quantum yield (0.89 versus 0.63) than the hydrolysis product of MUP.![]() The low pKa of its hydrolysis product makes DiFMUP a sensitive substrate for the continuous assay of acid phosphatases (Figure 10.3.4, Figure 10.3.5), which is not possible with MUP because its fluorescence must be measured at alkaline pH (pH >9) if the conventional detection configuration (excitation/emission = 360/460 nm) is used. Furthermore, with its high fluorescence quantum yield, DiFMUP increases the sensitivity of both acid and alkaline phosphatase measurements. As with our fluorinated fluorescein derivatives (Oregon Green dyes, Fluorescein, Oregon Green and Rhodamine Green Dyes—Section 1.5), fluorination renders the methylumbelliferone fluorophore much less susceptible to photobleaching, yet does not significantly affect the extinction coefficient or excitation/emission maxima. DiFMUP is available as a single 5 mg vial (D6567) or as a set of 10 vials, each containing 10 mg of the substrate (D22065) for high-throughput screening applications. DiFMUP is also used in our EnzChek Phosphatase Detection Kit (E12020), our RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kits (R22067) and our RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit (R33700), which are described below with our other phosphatase assay kits.
The low pKa of its hydrolysis product makes DiFMUP a sensitive substrate for the continuous assay of acid phosphatases (Figure 10.3.4, Figure 10.3.5), which is not possible with MUP because its fluorescence must be measured at alkaline pH (pH >9) if the conventional detection configuration (excitation/emission = 360/460 nm) is used. Furthermore, with its high fluorescence quantum yield, DiFMUP increases the sensitivity of both acid and alkaline phosphatase measurements. As with our fluorinated fluorescein derivatives (Oregon Green dyes, Fluorescein, Oregon Green and Rhodamine Green Dyes—Section 1.5), fluorination renders the methylumbelliferone fluorophore much less susceptible to photobleaching, yet does not significantly affect the extinction coefficient or excitation/emission maxima. DiFMUP is available as a single 5 mg vial (D6567) or as a set of 10 vials, each containing 10 mg of the substrate (D22065) for high-throughput screening applications. DiFMUP is also used in our EnzChek Phosphatase Detection Kit (E12020), our RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kits (R22067) and our RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit (R33700), which are described below with our other phosphatase assay kits.
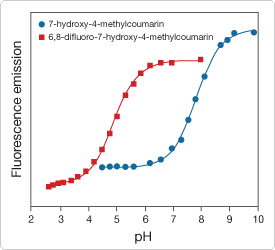
Figure 10.3.3 Comparison of the pH-dependent fluorescence responses of 7-hydroxy-4-methylcoumarin (H189) and 6,8-difluoro-7-hydroxy-4-methylcoumarin (DiFMU, D6566). Fluorescence intensities were measured for equal concentrations of the two dyes using excitation/emission at 355/460 nm. Under these conditions, DiFMU (pKa = 4.9) provides superior detection sensitivity in the 5–8 pH range compared to 7-hydroxy-4-methylcoumarin (pKa = 7.8).
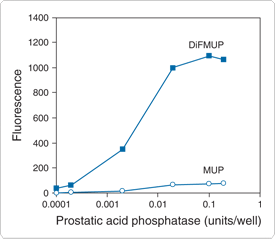
Figure 10.3.4 Comparison of DiFMUP (D6567, D22065) with MUP (M6491) for the detection of acid phosphatase activity at pH 5.5. Increasing amounts of prostatic acid phosphatase from human semen were reacted with 100 µM DiFMUP, the substrate in the EnzChek Acid Phosphatase Assay Kit (E12020), or 100 µM MUP, in 100 mM sodium acetate, pH 5.5, at room temperature. Fluorescence was measured after 60 minutes in a fluorescence microplate reader using excitation at 360 ± 20 nm and emission detection at 460 ± 20 nm.
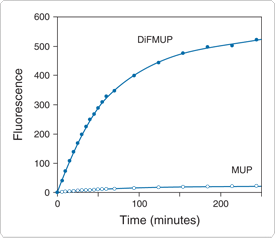
Figure 10.3.5 Time course of the reaction of prostatic acid phosphatase with DiFMUP (D6567, D22065) and MUP (M6491). Prostatic acid phosphatase from human semen (0.002 units) was reacted with 100 µM DiFMUP or 100 µM MUP in 100 mM sodium acetate, pH 5.5, at room temperature. Fluorescence was measured at the indicated times in a fluorescence microplate reader using excitation at 360 ± 20 nm and emission detection at 460 ± 20 nm.
Coumarin-Based Probe for Detection of Inorganic Phosphate
When conjugated to a mutant phosphate-binding protein, 7-diethylamino-3-((((2-maleimidyl)ethyl)amino)carbonyl)coumarin (MDCC, D10253; Thiol-Reactive Probes Excited with Ultraviolet Light—Section 2.3) has proven useful for direct, real-time measurement of inorganic phosphate release during enzymatic reactions.![]() An similar (7-diethylamino-3-((((2-iodoacetamido)ethyl)amino)carbonyl)coumarin (IDCC) conjugate of a mutant nucleoside diphosphate kinase has been used as a fluorescent sensor of the phosphorylation state of the kinase and to monitor purine nucleoside diphosphate concentrations in real time.
An similar (7-diethylamino-3-((((2-iodoacetamido)ethyl)amino)carbonyl)coumarin (IDCC) conjugate of a mutant nucleoside diphosphate kinase has been used as a fluorescent sensor of the phosphorylation state of the kinase and to monitor purine nucleoside diphosphate concentrations in real time.![]()
ELF 97 phosphate (E6588, E6589) yields a fluorescent precipitate at the site of phosphatase activity (![]() )—a process we call Enzyme-Labeled Fluorescence (ELF). Upon enzymatic cleavage (Figure 10.3.6), this weakly blue-fluorescent substrate yields an extremely photostable green-fluorescent precipitate that is up to 40 times brighter than the signal achieved when using either directly labeled fluorescent hybridization probes or fluorescent secondary detection methods in comparable applications.
)—a process we call Enzyme-Labeled Fluorescence (ELF). Upon enzymatic cleavage (Figure 10.3.6), this weakly blue-fluorescent substrate yields an extremely photostable green-fluorescent precipitate that is up to 40 times brighter than the signal achieved when using either directly labeled fluorescent hybridization probes or fluorescent secondary detection methods in comparable applications.![]() A particularly prominent application of ELF 97 phosphate is the detection of tartrate-resistant acid phosphatase (TRAP) activity, a marker of osteoclast differentiation from hematopoietic stem cells
A particularly prominent application of ELF 97 phosphate is the detection of tartrate-resistant acid phosphatase (TRAP) activity, a marker of osteoclast differentiation from hematopoietic stem cells ![]() (HSC). Using ELF 97 phosphate and confocal laser-scanning microscopy, researchers have developed a semiautomated method for analyzing the position within a regenerating newt limb of transfected cells expressing the secreted alkaline phosphatase reporter gene.
(HSC). Using ELF 97 phosphate and confocal laser-scanning microscopy, researchers have developed a semiautomated method for analyzing the position within a regenerating newt limb of transfected cells expressing the secreted alkaline phosphatase reporter gene.![]() Kits based on our ELF 97 phosphate include:
Kits based on our ELF 97 phosphate include:
These kits and their contents are described in detail in Enzyme-Labeled Fluorescence (ELF) Signal Amplification Technology—Section 6.3. ELF 97 phosphate is also available separately as a 5 mM solution in water containing sodium azide (E6589) or in a 0.2 µm–filtered solution (E6588). Filtration of ELF 97 phosphate through an ELF spin filter (E6606) is recommended before use. The ELF 97 alcohol (Figure 10.3.7) is available as a concentrated solution in DMSO (E6578, Introduction to Enzyme Substrates and Their Reference Standards—Section 10.1).
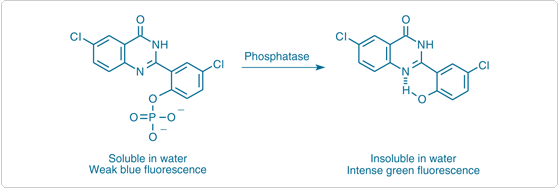
Figure 10.3.6 Principle of enzyme-mediated formation of the fluorescent ELF 97 alcohol precipitate from the ELF 97 phosphatase substrate.
BODIPY FL ATP-γ-S and BODIPY FL GTP-γ-S Thioesters: Substrates for the Fhit Nucleotide-Binding Protein
BODIPY FL ATP-γ-S (A22184) and BODIPY FL GTP-γ-S (G22183) thioesters are important substrates for Fhit (Figure 10.3.8), a member of the histidine triad superfamily of nucleotide-binding proteins that binds and cleaves diadenosine polyphosphates.![]() Fhit, one of the most frequently inactivated proteins in lung cancer, functions as a tumor suppressor by inducing apoptosis.
Fhit, one of the most frequently inactivated proteins in lung cancer, functions as a tumor suppressor by inducing apoptosis.![]() In addition to their use for screening potential Fhit inhibitors, these BODIPY FL nucleotides should also be important for studying other nucleotide-binding proteins because they bind to ATP- or GTP-binding sites but are not metabolized (Probes for Protein Kinases, Protein Phosphatases and Nucleotide-Binding Proteins—Section 17.3). As with other fluorescent nucleotides, the fluorescence polarization properties of the bound nucleotide should differ from that of the free nucleotide. The fluorescence of BODIPY FL GTP-γ-S thioester is quenched ~90% relative to that of the free dye (Figure 10.3.9) but is recovered upon binding to at least some G-proteins.
In addition to their use for screening potential Fhit inhibitors, these BODIPY FL nucleotides should also be important for studying other nucleotide-binding proteins because they bind to ATP- or GTP-binding sites but are not metabolized (Probes for Protein Kinases, Protein Phosphatases and Nucleotide-Binding Proteins—Section 17.3). As with other fluorescent nucleotides, the fluorescence polarization properties of the bound nucleotide should differ from that of the free nucleotide. The fluorescence of BODIPY FL GTP-γ-S thioester is quenched ~90% relative to that of the free dye (Figure 10.3.9) but is recovered upon binding to at least some G-proteins.![]()
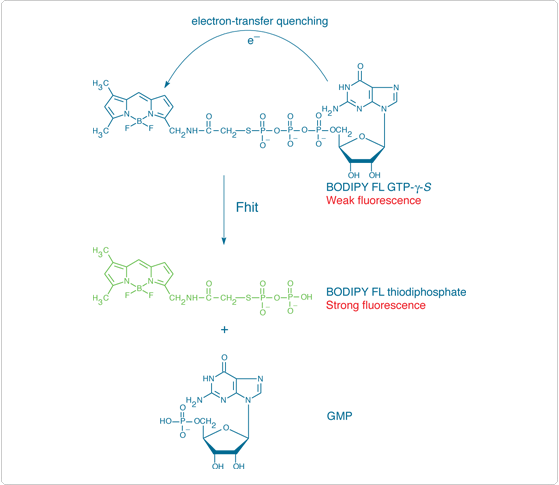
Figure 10.3.8 Principle of fluorescence-based detection of the diadenosine triphosphate hydrolase activity of Fhit using BODIPY FL GTP-γ-S thioester (G22183) as a substrate analog.
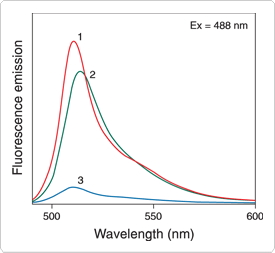
Figure 10.3.9 Fluorescence emission spectra of (1) free BODIPY FL dye in phosphate-buffered saline, pH 7.2; (2) BODIPY FL ATP (A12410); and (3) BODIPY FL GTP (G12411). Samples were prepared with equal absorbance at the excitation wavelength (488 nm). The areas under the curves are therefore proportional to the relative fluorescence quantum yields, clearly showing the quenching effect caused by interaction of the BODIPY FL fluorophore with the guanine base of GTP.
NovaBright Secreted Placental Alkaline Phosphatase (SEAP) Enzyme Reporter Gene Chemiluminescent Detection System
The NovaBright Secreted Placental Alkaline Phosphatase (SEAP) Enzyme Reporter Gene Chemiluminescent Detection System is a chemiluminescent reporter gene assay designed for the rapid and sensitive detection of secreted placental alkaline phosphatase (SEAP) in cell culture media. SEAP, a truncated form of human placental alkaline phosphatase (PLAP), is commonly used as a reporter protein that is secreted into the cell culture media and detected by testing aliquots of media, leaving cells intact for further experimentation. Detection of nonsecreted placental alkaline phosphatase is also possible using the NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System.
The NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System employs the CSPD chemiluminescent substrate and Emerald luminescence enhancer to achieve sensitive SEAP detection with a wide dynamic range.![]() In this NovaBright assay, SEAP activity is measure 48–72 hours after cell transfection by first incubating the cell culture medium or cell lysate with a buffer that selectively inhibits nonplacental alkaline phosphatase (serum and endogenous cellular alkaline phosphatases), and then with CSPD-containing reaction buffer until maximum light emission is reached (approximately 20 minutes). The light emission kinetics provide a persistent glow signal that enables measurement over a wide time interval. Light signal output is measured in a luminometer, without the need for automated injection capability. The NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System has been used to detect SEAP activity in cell culture media and to quantitate nonsecreted placental alkaline phosphatase in both cell and tissue extracts.
In this NovaBright assay, SEAP activity is measure 48–72 hours after cell transfection by first incubating the cell culture medium or cell lysate with a buffer that selectively inhibits nonplacental alkaline phosphatase (serum and endogenous cellular alkaline phosphatases), and then with CSPD-containing reaction buffer until maximum light emission is reached (approximately 20 minutes). The light emission kinetics provide a persistent glow signal that enables measurement over a wide time interval. Light signal output is measured in a luminometer, without the need for automated injection capability. The NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System has been used to detect SEAP activity in cell culture media and to quantitate nonsecreted placental alkaline phosphatase in both cell and tissue extracts.
Chemiluminescent reporter assays for secreted placental alkaline phosphatase may be conducted in cells that have endogenous nonplacental alkaline phosphatase activity. Endogenous nonplacental enzyme activity is significantly reduced with a combination of heat inactivation and differential inhibitors that do not significantly inhibit the transfected placental alkaline phosphatase. It is important to determine the level of endogenous enzyme in media of nontransfected cells in order to establish assay background. Certain cell lines, such as HeLa and others derived from cervical cancers, may express placental alkaline phosphatase, which will likely produce high assay backgrounds when shed into the media, and therefore we do not recommend the use of secreted alkaline phosphatase as a reporter system in these cell lines.
Each NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System provides:
- CSPD substrate
- Purified human placental alkaline phosphatase
- Dilution buffer
- Assay buffer, containing a mixture of nonplacental alkaline phosphatase inhibitors
- Reaction buffer, containing the Emerald luminescence enhancer
- Detailed protocols (NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System)
Sufficient reagents are supplied for 400 (N10559) or 1200 (N10560) microplate assays based on the protocol provided.
NovaBright Secreted Placental Alkaline Phosphatase (SEAP) Enzyme Reporter Gene Chemiluminescent Detection System 2.0
The next-generation NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System 2.0 employs CSPD chemiluminescent substrate and an advanced Emerald-III luminescence enhancer that produces higher signal intensity and higher assay signal/noise performance than the original assay system. In addition, all the reagents in the new assay system are provided ready-to-use without the need for any reagent preparation steps, allowing fewer assay steps and a shorter assay duration. Depending on the expression system, secreted placental alkaline phosphatase can be measured in as little as 6 hours after cell transfection.
In this assay, cell culture medium is incubated first with an assay buffer that contains selective inhibitors of nonplacental alkaline phosphatase (serum and endogenous cellular alkaline phosphatases) and then with the reaction buffer containing CSPD substrate and Emerald-III light emission enhancer until maximum light signal is reached in approximately 20 minutes. The light emission kinetics provide a persistent glow signal, with light emission half-life of approximately 2.5 hours, enabling measurement over a wide time interval. Light emission is measured in a luminometer without the need for automated injection capability. As with the original system, we do not recommend the use of this assay in cell lines such as HeLa and others derived from cervical cancers, that may express placental alkaline phosphatase.
Each NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System 2.0 provides:
- Reaction buffer, containing CSPD substrate and Emerald-III luminescence enhancer
- Purified human placental alkaline phosphatase
- Assay buffer, containing a mixture of nonplacental alkaline phosphatase inhibitors
- Detailed protocols (NovaBright SEAP Enzyme Reporter Gene Chemiluminescent Detection System 2.0)
Sufficient reagents are supplied for 192 (N10577) or 960 (N10578) microplate assays based on the protocol provided.
Chemiluminescent Alkaline Phosphatase ELISA Sampler Kit
The Chemiluminescent Alkaline Phosphatase ELISA Kits employ CSPD or CDP-Star 1,2-dioxetane substrates for alkaline phosphatase with Sapphire-II or Emerald‑II enhancer in a system designed for rapid and ultrasensitive analyte detection in enzyme-linked immunoassays.![]() Maximum light emission from alkaline phosphatase-activated CSPD or CDP-Star substrate is reached in 5 to 60 minutes, depending on the temperature and the substrate chosen. Enzymatic dephosphorylation of substrate occurs at a constant rate proportional to enzyme concentration; the resulting phenolate anion decomposes with a finite half-life (Figure 10.3.10). Light emission can be quantitated with a variety of luminometers without the need for solution injection.
Maximum light emission from alkaline phosphatase-activated CSPD or CDP-Star substrate is reached in 5 to 60 minutes, depending on the temperature and the substrate chosen. Enzymatic dephosphorylation of substrate occurs at a constant rate proportional to enzyme concentration; the resulting phenolate anion decomposes with a finite half-life (Figure 10.3.10). Light emission can be quantitated with a variety of luminometers without the need for solution injection.
Enzyme-linked immunosorbent assays (ELISAs) can be formatted in several configurations on a variety of solid supports including microplate wells, tubes, polystyrene beads or ferrite particles. The high sensitivity obtained with 1,2-dioxetane substrates is demonstrated in a sandwich immunoassay format that employs a biotinylated detector antibody and streptavidin alkaline phosphatase conjugate for quantitating recombinant human IL-6 (rhIL-6). The results obtained with CSPD substrate/Sapphire-II enhancer (Figure 10.3.11) show a significant improvement in signal-to-noise performance at all concentrations of rhIL-6 and a much wider assay dynamic range compared to those obtained with the fluorescent substrate 4-methylumbelliferyl phosphate (MUP, M6491), and the colorimetric substrate p-nitrophenyl phosphate ![]() (pNPP).
(pNPP).
We offer four different Chemiluminescent Alkaline Phosphatase ELISA Kits, as well as a Chemiluminescent Alkaline Phosphatase ELISA Sampler Kit. Each kit provides concentrated assay buffer and blocking reagent, as well as one of the following substrate/enhancer solutions:
The Chemiluminescent Alkaline Phosphatase ELISA Sampler Kit (C10556) provides assay buffer, blocking reagent and sample sizes of all four substrate/enhancer solutions. All five kits provide sufficient reagents for 1000 assays based on the provided protocol.
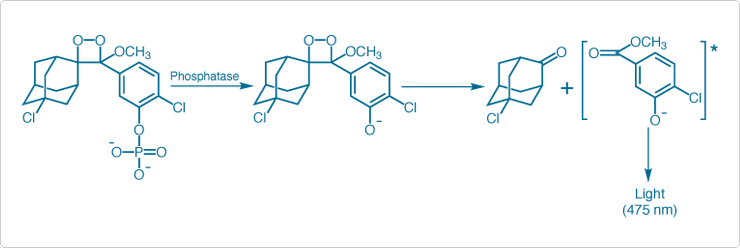
Figure 10.3.10 Mechanism of phosphatase-dependent chemiluminescence generation by CDP-Star substrate. Luminescence emission is shifted to 461 nm or 542 nm respectively by the Sapphire-II and Emerald-II enhancers.
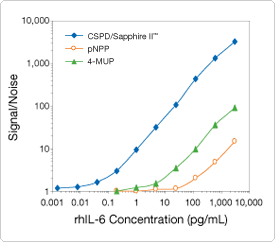
Figure 10.3.11 Comparison of detection sensitivities for the ELISA quantitation of recombinant human IL-6 (rhIL-6) using the chemiluminescent phosphatase substrate/enhancer in the Chemiluminescent Alkaline Phosphatase ELISA Kit #1 (CSPD substrate/Sapphire-II enhancer in C10552), the fluorescent phosphatase substrate 4-methylumbelliferyl phosphate (MUP, M6491) and the colorimetric phosphatase substrate p-nitrophenyl phosphate (pNPP).
NBT/BCIP Reagent Kit
5-Bromo-4-chloro-3-indolyl phosphate (BCIP) is commonly used with a number of different chromogens in various histological and molecular biology techniques. Hydrolysis of this indolyl phosphate, followed by oxidation, produces a blue-colored precipitate at the site of enzymatic activity. For convenience, we offer the NBT/BCIP Reagent Kit (N6547), which provides 1 g samples of BCIP as well as its co-precipitant NBT.
NBT: A Co-Precipitant for the BCIP Reaction
Nitro blue tetrazolium (NBT, N6495) is the most commonly used electron-transfer agent and co-precipitant for the BCIP reaction, forming a dark blue, precisely localized precipitate in the presence of alkaline phosphatase ![]() (Figure 10.3.12,
(Figure 10.3.12, ![]() ).
).
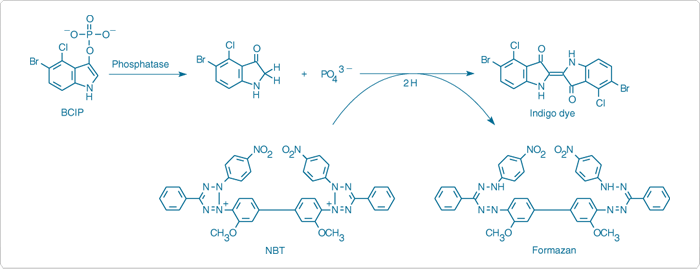
Figure 10.3.12 Principle of enzyme-linked detection using the reagents in our NBT/BCIP Reagent Kit (N6547). Phosphatase hydrolysis of BCIP is coupled to reduction of NBT, yielding a formazan and an indigo dye that together form a black-purple–colored precipitate.
We have developed some unique products for following the activity of phosphatases, polymerases and nucleases. Our PiPer Phosphate and PiPer Pyrophosphate Assay Kits provide ultrasensitive assays for free phosphate and pyrophosphate, respectively, through the formation of resorufin, which can be detected either fluorometrically or spectrophotometrically. Our EnzChek Phosphate and EnzChek Pyrophosphate Assay Kits provide spectrophotometric assays for inorganic phosphate and pyrophosphate, respectively. Both the PiPer and the EnzChek Phosphate Assay Kits permit continuous measurement of the activity of ATPases, GTPases, phosphatases, nucleotidases and a number of enzymes that produce or consume inorganic phosphate. Moreover, these enzyme assay kits utilize the natural substrate for the enzyme rather than a synthetic analog and detect the inorganic phosphate produced using enzyme-coupled reactions. The PiPer Kit is more sensitive and better suited for high-throughput studies in microplates, but requires an additional enzymatic step, whereas the EnzChek Kit is less sensitive but has a simpler protocol. In both the PiPer and the EnzChek Pyrophosphate Assay Kits, the phosphate assay is coupled with the enzyme, pyrophosphatase, in order to monitor the activity of pyrophosphate-producing enzymes such as DNA- and RNA-polymerases, adenylate cyclase and guanylate cyclase.![]()
In addition to these kits, our RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kit (R22067) and RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit (R33700) utilize DiFMUP for assaying tyrosine phosphatase activity and serine/threonine phosphatase activity, respectively, as well as for screening phosphatase inhibitors in a convenient high-throughput format.
PiPer Phosphate Assay Kit
The PiPer Phosphate Assay Kit (P22061) provides an ultrasensitive assay that detects free phosphate in solution through formation of the fluorescent product resorufin. Because resorufin also has strong absorption, the assay can be performed either fluorometrically or spectrophotometrically. This kit can be used to detect inorganic phosphate (Pi) in a variety of samples or to monitor the kinetics of phosphate release by a variety of enzymes, including ATPases, GTPases, 5'-nucleotidase, protein phosphatases, acid and alkaline phosphatases and phosphorylase kinase. Furthermore, the assay can be modified to detect virtually any naturally occurring organic phosphate molecule by including an enzyme that can specifically digest the organic phosphate to liberate inorganic phosphate.
In the PiPer phosphate assay (Figure 10.3.13), maltose phosphorylase converts maltose (in the presence of Pi) to glucose 1-phosphate and glucose. Then glucose oxidase converts the glucose to gluconolactone and H2O2. Finally, with horseradish peroxidase as a catalyst, the H2O2 reacts with the Amplex Red reagent (10-acetyl-3,7-dihydroxyphenoxazine) to generate resorufin, which has absorption/emission maxima of ~571/585 nm.![]() The resulting increase in fluorescence or absorption is proportional to the amount of Pi in the sample. This kit can be used to detect as little as 0.2 µM Pi by fluorescence (Figure 10.3.14) or 0.4 µM Pi by absorption.
The resulting increase in fluorescence or absorption is proportional to the amount of Pi in the sample. This kit can be used to detect as little as 0.2 µM Pi by fluorescence (Figure 10.3.14) or 0.4 µM Pi by absorption.
Each PiPer Phosphate Assay Kit contains:
- Amplex Red reagent
- Dimethylsulfoxide (DMSO)
- Concentrated reaction buffer
- Recombinant maltose phosphorylase from Escherichia coli
- Maltose
- Glucose oxidase from Aspergillus niger
- Horseradish peroxidase
- Phosphate standard
- Hydrogen peroxide
- Detailed protocols for detecting phosphatase activity (PiPer Phosphate Assay Kit)
Sufficient reagents are provided for approximately 1000 assays using a reaction volume of 100 µL per assay and either a fluorescence or absorbance microplate reader.
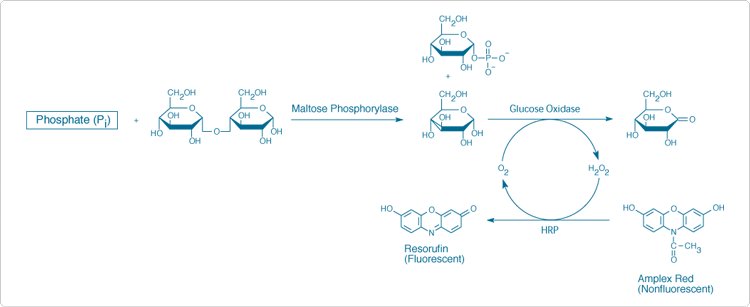
Figure 10.3.13 Principle of the PiPer Phosphate Assay Kit (P22061). In the presence of inorganic phosphate, maltose phosphorylase converts maltose to glucose 1-phosphate and glucose. Then, glucose oxidase converts the glucose to gluconolactone and H2O2. Finally, with horseradish peroxidase (HRP) as a catalyst, the H2O2 reacts with the Amplex Red reagent to generate the highly fluorescent resorufin. The resulting increase in fluorescence or absorption is proportional to the amount of Pi in the sample.
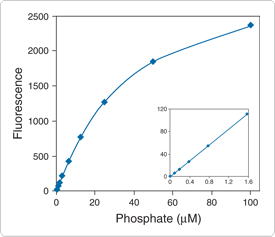
Figure 10.3.14 Detection of inorganic phosphate using the PiPer Phosphate Assay Kit (P22061). Each reaction contained 50 µM Amplex Red reagent, 2 U/mL maltose phosphorylase, 1 mM maltose, 1 U/mL glucose oxidase and 0.2 U/mL HRP in 1X reaction buffer. Reactions were incubated at 37°C. After 60 minutes, fluorescence was measured in a fluorescence microplate reader using excitation at 530 ± 12.5 nm and fluorescence detection at 590 ± 17.5 nm. Data points represent the average of duplicate reactions, and a background value of 43 (arbitrary units) was subtracted from each reading
PiPer Pyrophosphate Assay Kit
The PiPer Pyrophosphate Assay Kit (P22062) provides a sensitive fluorometric or colorimetric method for measuring the inorganic pyrophosphate (PPi) in experimental samples or for monitoring the kinetics of PPirelease by a variety of enzymes, including DNA and RNA polymerases, adenylate cyclase and S-acetyl coenzyme A synthetase. In the PiPer pyrophosphate assay, inorganic pyrophosphatase hydrolyzes PPi to two molecules of inorganic phosphate (Pi). The Pi then enters into the same cascade of reactions as it does in the PiPer Phosphate Assay Kit (Figure 10.3.13). In this case, the resulting increase in fluorescence or absorption is proportional to the amount of PPi in the sample. This kit can be used to detect as little as 0.1 µM PPi by fluorescence or 0.2 µM PPi by absorption (Figure 10.3.15).
Each PiPer Pyrophosphate Assay Kit contains:
- Amplex Red reagent
- Dimethylsulfoxide (DMSO)
- Concentrated reaction buffer
- Recombinant maltose phosphorylase from Escherichia coli
- Maltose
- Glucose oxidase from Aspergillus niger
- Horseradish peroxidase
- Inorganic pyrophosphatase from baker's yeast
- Pyrophosphate standard
- Detailed protocols for detecting pyrophosphatase activity (PiPer Pyrophosphate Assay Kit)
Sufficient reagents are provided for approximately 1000 assays using a reaction volume of 100 µL per assay and either a fluorescence or absorbance microplate reader.
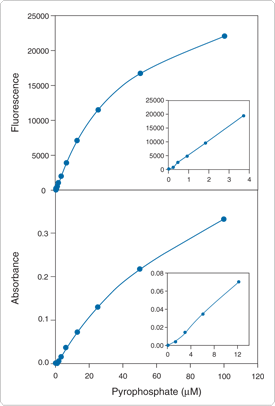
Figure 10.3.15 Detection of pyrophosphate using the PiPer Pyrophosphate Assay Kit (P22062). Each reaction contained 50 µM Amplex Red reagent, 0.01 U/mL inorganic pyrophosphatase, 2 U/mL maltose phosphorylase, 0.2 mM maltose, 1 U/mL glucose oxidase and 0.2 U/mL HRP in 1X reaction buffer. Reactions were incubated at 37°C. After 60 minutes, A) fluorescence was measured in a fluorescence-based microplate reader using excitation at 530 ± 12.5 nm and fluorescence detection at 590 ± 17.5 nm or B) absorbance was measured in an absorption-based microplate reader at 576 ± 5 nm. Data points represent the average of duplicate reactions. In panel A, a background value of 78 (arbitrary units) was subtracted from each reading; in panel B, a background absorbance of 0.011 was subtracted from each reading.
EnzChek Phosphate Assay Kit
The EnzChek Phosphate Assay Kit (E6646), which is based on a method originally described by Webb,![]() provides an easy enzymatic assay for detecting Pi from multiple sources through formation of a chromophoric product (Figure 10.3.16). Although this kit is usually used to determine the Pi produced by a wide variety of enzymes such as ATPases, kinases and phosphatases, it can also be used to specifically quantitate Pi with a sensitivity of ~2 µM Pi (~0.2 µg/mL) (Figure 10.3.17). Moreover, this spectrophotometric assay has proven useful for determining the level of Pi contamination in the presence of high concentrations of acid-labile phosphates using a microplate reader.
provides an easy enzymatic assay for detecting Pi from multiple sources through formation of a chromophoric product (Figure 10.3.16). Although this kit is usually used to determine the Pi produced by a wide variety of enzymes such as ATPases, kinases and phosphatases, it can also be used to specifically quantitate Pi with a sensitivity of ~2 µM Pi (~0.2 µg/mL) (Figure 10.3.17). Moreover, this spectrophotometric assay has proven useful for determining the level of Pi contamination in the presence of high concentrations of acid-labile phosphates using a microplate reader.![]() Because the sulfate anion competes with Pi for binding to purine nucleoside phosphorylase (PNP), this kit can be adapted for measurement of sulfate concentrations between 0.1 and 10 mM in the presence of a low (<100 µM) fixed Pi concentration.
Because the sulfate anion competes with Pi for binding to purine nucleoside phosphorylase (PNP), this kit can be adapted for measurement of sulfate concentrations between 0.1 and 10 mM in the presence of a low (<100 µM) fixed Pi concentration.![]()
Each EnzChek Phosphate Assay Kit contains:
- 2-Amino-6-mercapto-7-methylpurine riboside (MESG)
- Purine nucleoside phosphorylase (PNP)
- Concentrated reaction buffer
- KH2PO4 standard
- Detailed protocols for detecting and quantitating Pi (EnzChek Phosphate Assay Kit)
In the presence of Pi, MESG is enzymatically converted by PNP to ribose 1-phosphate and the chromophoric product 2-amino-6-mercapto-7-methylpurine (Figure 10.3.16). This kit contains sufficient reagents for about 100 phosphate assays using 1 mL assay volumes and standard cuvettes. The substrate MESG and the enzyme PNP included in our EnzChek Phosphate Assay Kit have been adapted for monitoring the kinetics of phosphate release by:
- Actin-activated myosin ATPase

- Actin polymerization

- Aminoacyl-tRNA synthetase

- Aspartate transcarbamylase

- ArsA ATPase, the catalytic subunit of the arsenite pump

- Dethiobiotin synthetase

- Glycerol kinase

- Glycogen phosphorylase

- GTPases

- ATPases

- myo-Inositol monophosphatase

- Phospholysine and phosphohistidine phosphatases

- Phosphorylase a phosphatase

- Phosphorylase kinase

- Serine phosphatase

- Self-assembly of actin and tubulin

- Autodephosphorylation of CheY, a response regulator that mediates bacterial chemotaxis

Moreover, the EnzChek phosphate assay is sufficiently fast and quantitative to permit stopped-flow kinetic experiments on enzymes that produce phosphate, an important development for mechanistic enzyme studies.![]() Although this kit is usually used to determine the inorganic phosphate produced by enzymes such as ATPases and GTPases, it can also be used to specifically quantitate inorganic phosphate contamination of reagents and solution, with a detection limit of ~2 µM Pi (~0.2 µg/mL) and an effective range between 2 and 150 µM inorganic phosphate (between 2 and 150 nanomoles phosphate in a 1 mL volume). For example, the assay has been used for the rapid assay of inorganic phosphate in the presence of high concentrations of acid-labile phosphates using a microplate reader.
Although this kit is usually used to determine the inorganic phosphate produced by enzymes such as ATPases and GTPases, it can also be used to specifically quantitate inorganic phosphate contamination of reagents and solution, with a detection limit of ~2 µM Pi (~0.2 µg/mL) and an effective range between 2 and 150 µM inorganic phosphate (between 2 and 150 nanomoles phosphate in a 1 mL volume). For example, the assay has been used for the rapid assay of inorganic phosphate in the presence of high concentrations of acid-labile phosphates using a microplate reader.![]() The reagents in this kit can also be used as a phosphate "mop" to remove almost all inorganic phosphate from a protein solution.
The reagents in this kit can also be used as a phosphate "mop" to remove almost all inorganic phosphate from a protein solution.![]()
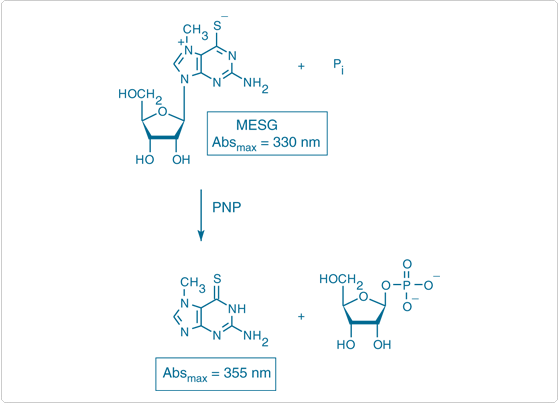
Figure 10.3.16 Enzymatic conversion of 2-amino-6-mercapto-7-methylpurine riboside (MESG) to ribose 1-phosphate and 2-amino-6-mercapto-7-methylpurine by purine nucleoside phosphorylase (PNP), reagents supplied in the EnzChek Phosphate Assay Kit (E6646). The accompanying change in the absorption maximum (Abs) allows quantitation of inorganic phosphate (Pi) consumed in the reaction.
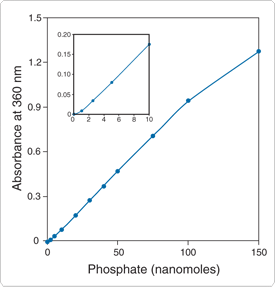
Figure 10.3.17 Quantitative analysis of inorganic phosphate using the EnzChek Phosphate Assay Kit (E6646). KH2PO4 was used as the source for the inorganic phosphate, and the absorbance at 360 nm was corrected for background absorbance. The inset shows an enlargement of the standard curve, demonstrating the lower range of the assay; the units are the same.
EnzChek Pyrophosphate Assay Kit
In the EnzChek Pyrophosphate Assay Kit (E6645), we have adapted the method provided in the EnzChek Phosphate Assay Kit to permit the sensitive spectrophotometric detection of PPi, which is converted by the enzyme pyrophosphatase to Pi.![]() Because two moles of Pi are released per mole of PPi consumed, the sensitivity limit of the EnzChek Pyrophosphate Assay Kit is 1 µM PPi (~0.2 µg/mL). This assay has been modified to continuously detect several enzymes that liberate PPi
Because two moles of Pi are released per mole of PPi consumed, the sensitivity limit of the EnzChek Pyrophosphate Assay Kit is 1 µM PPi (~0.2 µg/mL). This assay has been modified to continuously detect several enzymes that liberate PPi ![]() such as aminoacyl-tRNA synthetase,
such as aminoacyl-tRNA synthetase,![]() luciferase, cytidylyl transferase,
luciferase, cytidylyl transferase,![]() S-acetyl coenzyme A synthetase,
S-acetyl coenzyme A synthetase,![]() adenylate cyclase and guanylyl cyclase.
adenylate cyclase and guanylyl cyclase.![]()
Each EnzChek Pyrophosphate Assay Kit contains:
- Inorganic pyrophosphatase
- 2-Amino-6-mercapto-7-methylpurine riboside (MESG)
- Purine nucleoside phosphorylase (PNP)
- Concentrated reaction buffer
- Na2P2O7 standard
- Detailed protocols for detecting and quantitating PPi (EnzChek Pyrophosphate Assay Kit)
This kit contains sufficient reagents for about 100 PPi assays using standard 1 mL assay volumes and standard cuvettes.
EnzChek Phosphatase Assay Kit
The EnzChek Phosphatase Assay Kit ![]() (E12020), which includes the DiFMUP substrate, can be used to continuously detect phosphatase activity at neutral, basic or moderately acidic pH. Using the assay outlined in the kit protocol, we have found that DiFMUP is about 100 times more sensitive than MUP for the detection of prostatic acid phosphatase at pH 5.5 (Figure 10.3.4) and can measure this activity at a pH that is optimal for the enzyme (Figure 10.3.5). The EnzChek Phosphatase Assay Kit is perfect for the continuous assay of prostatic acid phosphatase, protein phosphatase 1 or almost any other phosphatase that can be assayed with nonprotein-based substrates such as MUP or 4-nitrophenyl phosphate (pNPP). Each EnzChek Phosphatase Assay Kit contains:
(E12020), which includes the DiFMUP substrate, can be used to continuously detect phosphatase activity at neutral, basic or moderately acidic pH. Using the assay outlined in the kit protocol, we have found that DiFMUP is about 100 times more sensitive than MUP for the detection of prostatic acid phosphatase at pH 5.5 (Figure 10.3.4) and can measure this activity at a pH that is optimal for the enzyme (Figure 10.3.5). The EnzChek Phosphatase Assay Kit is perfect for the continuous assay of prostatic acid phosphatase, protein phosphatase 1 or almost any other phosphatase that can be assayed with nonprotein-based substrates such as MUP or 4-nitrophenyl phosphate (pNPP). Each EnzChek Phosphatase Assay Kit contains:
- DiFMUP substrate
- Concentrated reaction buffer
- Acid phosphatase from potato for use as a positive control
- 6,8-Difluoro-7-hydroxy-4-methylcoumarin for use as a reference standard
- Detailed protocols for detecting phosphatase activity (EnzChek Phosphatase Assay Kit)
The kit provides sufficient reagents for performing approximately 1000 assays, using a reaction volume of 100 µL per assay.
RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kits
Protein tyrosine phosphatases (PTP) represent a large family of enzymes that play a very important role in intra- and intercellular signaling. PTPs work antagonistically with protein tyrosine kinases to regulate signal transduction pathways in response to a variety of signals, including hormones and mitogens.![]() Our RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kit (R22067) provide researchers with a sensitive and convenient method to monitor PTP and screen PTP inhibitors in a variety of research areas
Our RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kit (R22067) provide researchers with a sensitive and convenient method to monitor PTP and screen PTP inhibitors in a variety of research areas ![]() (Figure 10.3.18).
(Figure 10.3.18).
The EnzChek tyrosine phosphatase assay is based on 6,8-difluoro-4-methylumbelliferyl phosphate ![]() (DiFMUP; D6567, D22065). The EnzChek tyrosine phosphatase assay is continuous, allowing researchers to easily measure fluorescence at various time points in order to follow the kinetics of the reaction. Furthermore, the assay is not affected by free phosphate and is compatible with most nonionic detergents, resulting in minimal sample processing before analysis. Most importantly, each assay well contains inhibitors to ensure that the assay is selective for tyrosine phosphatases; other phosphatases, including serine/threonine phosphatases, will not hydrolyze DiFMUP under our assay conditions (Figure 10.3.19). Unlike phosphopeptide-based assays, this DiFMUP-based assay can be used to monitor a variety of tyrosine phosphatases, including PTP-1B and CD-45 (Figure 10.3.19). Tyrosine phosphatase inhibitors can be evaluated quantitatively in the assay for their effect on tyrosine phosphatase activity.
(DiFMUP; D6567, D22065). The EnzChek tyrosine phosphatase assay is continuous, allowing researchers to easily measure fluorescence at various time points in order to follow the kinetics of the reaction. Furthermore, the assay is not affected by free phosphate and is compatible with most nonionic detergents, resulting in minimal sample processing before analysis. Most importantly, each assay well contains inhibitors to ensure that the assay is selective for tyrosine phosphatases; other phosphatases, including serine/threonine phosphatases, will not hydrolyze DiFMUP under our assay conditions (Figure 10.3.19). Unlike phosphopeptide-based assays, this DiFMUP-based assay can be used to monitor a variety of tyrosine phosphatases, including PTP-1B and CD-45 (Figure 10.3.19). Tyrosine phosphatase inhibitors can be evaluated quantitatively in the assay for their effect on tyrosine phosphatase activity.
Each RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kit (R22067) includes:
- One RediPlate 96 EnzChek tyrosine phosphatase assay 96-well microplate
- Reaction buffer
- Detailed assay protocols (RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kit)
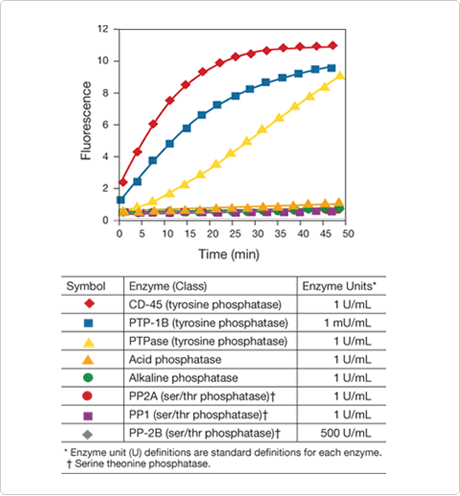
Figure 10.3.19 Specificity of the RediPlate 96 EnzChek Tyrosine Phosphatase Assay Kit (R22067). The phosphatases listed in the tables were applied to a RediPlate 96 EnzChek tyrosine phosphatase assay microplate. At the indicated time points, the fluorescence was measured in a fluorescence microplate reader using excitation at 355 ± 20 nm and emission at 460 ± 12.5 nm.
RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit
The majority of protein phosphorylation occurs on serine and threonine residues, with <0.01–0.05% on tyrosine residues. Serine/threonine phosphatases represent a large family of enzymes that have been implicated in the regulation of metabolism, transcription, translation, differentiation, cell cycle, cytoskeletal dynamics, oncogenesis and signal transduction. The RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit (R33700) provides a fast, simple and direct fluorescence-based assay for detecting serine/threonine phosphatases and their corresponding modulators and inhibitors ![]() (Figure 10.3.18).
(Figure 10.3.18).
As with the RediPlate 96 EnzChek Tyrosine Phosphatase Kit, the substrate incorporated in the RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit is DiFMUP. Inhibitors are included in each assay well to ensure that the assay is selective for serine/threonine phosphatases; under the prescribed assay conditions, other phosphatases, including tyrosine phosphatases, do not significantly react with the substrate (Figure 10.3.20). Furthermore, unlike phosphopeptide-based assays, this DiFMUP-based assay can be used to monitor a variety of serine/threonine phosphatases including PP-1, PP-2A and PP-2B (Figure 10.3.20). Serine/threonine phosphatase inhibitors can be evaluated quantitatively in the assay for their effect on serine/threonine phosphatase activity (Figure 10.3.21). Additional advantages of this RediPlate assay include compatibility with nonionic detergents and insensitivity to free phosphate, resulting in minimal sample processing before analysis.
Each RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit includes:
- One RediPlate 96 EnzChek serine/threonine phosphatase assay 96-well microplate
- Concentrated reaction buffer
- NiCl2
- MnCl2
- Dithiothreitol
- Detailed assay protocols (RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit)
To ensure the integrity of the pre-dispensed reagents, the 96-well microplate provided in both RediPlate Protein Phosphatase Assay Kits is packaged in a resealable foil pouch and consists of twelve removable strips, each with eight wells (Figure 10.3.22). Eleven of the strips (88 wells) are preloaded with the fluorogenic substrate DiFMUP; the remaining strip, marked with black tabs, contains a dilution series of the DiFMU reference standard for generating a standard curve.
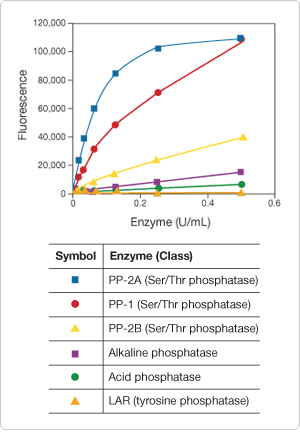
Figure 10.3.20 Specificity of the RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit (R33700) for serine/threonine phosphatases. The phosphatases listed in the tables were applied at the indicated concentrations to a RediPlate 96 EnzChek serine/threonine phosphatase assay microplate. Reactions were incubated at 37°C. After 1 hour, fluorescence was measured in a fluorescence microplate reader using excitation at 355 ± 20 nm and emission at 460 ± 12.5 nm.
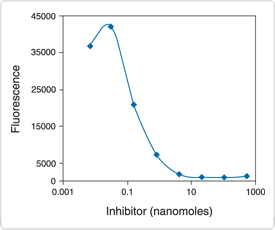
Figure 10.3.21 Detection of PP-2A inhibition by okadaic acid using the RediPlate 96 EnzChek Serine/Threonine Phosphatase Assay Kit (R33700). Each reaction contained 50 µM DiFMUP, 10 mU/mL PP-2A and the indicated concentration (log scale) of okadaic acid in reaction buffer containing 50 mM Tris-HCl, 0.1 mM CaCl2, 1 mM NiCl2, 125 µg/mL bovine serum albumin (BSA) and 0.05% Tween 20. Reactions were incubated at 37°C. After 30 minutes, fluorescence was measured in a fluorescence microplate reader using excitation at 355 ± 20 nm and emission at 460 ± 12.5 nm.

Figure 10.3.22 A RediPlate 96 microplate
EnzChek Ultra Phytase Assay Kit
Phytases catalyze the sequential hydrolysis of phytate (myoinositol hexakis phosphate; phytin; phytic acid) to less phosphorylated myo-inositol compounds and inorganic phosphate. The EnzChek Ultra Phytase Assay Kit (E33701) utilizes a series of linked enzymatic reactions for the detection of phytase activity based on the measurement of phosphate released from the substrate phytic acid (Figure 10.3.23). In the initial step, phytase catalyzes the release of inorganic phosphate from phytic acid; in the presence of Pi, maltose phosphorylase converts maltose to glucose 1-phosphate and glucose. Glucose oxidase then converts the glucose to gluconolactone and H2O2. Finally, with horseradish peroxidase (HRP) as a catalyst, the H2O2reacts with the Amplex UltraRed reagent to generate a brightly fluorescent and strongly absorbing reaction product (excitation/emission maxima ~568/581 nm). The resulting increase in fluorescence is proportional to the amount of Pi in the sample. This relative measure of phytase activity can be used as the basis of a standard curve of phytase enzyme standards with known activity. The assay takes one hour at 37°C; under these conditions, the limit of detection is 0.001 FTU/mL and the intra-assay CV is ≤10%. The signal is not linear but can cover a two-log concentration range for phytase (Figure 10.3.24).
Each EnzChek Ultra Phytase Assay Kit provides:
- Amplex UltraRed reagent
- Dimethylsulfoxide (DMSO)
- Concentrated reaction buffer
- Sodium acetate, pH 5.5
- Phytic acid
- Maltose phosphorylase, recombinant from Escherichia coli (1 unit = the amount of enzyme that will convert maltose, in the presence of inorganic phosphate, to 1.0 mole of D-glucose and D-glucose 1-phosphate per minute at 37°C, pH 7.0)
- Glucose oxidase from Aspergillus niger (1 unit = the amount of enzyme that will oxidize 1.0 mole of -D-glucose to D-gluconolactone and H2O2 per minute at 35°C, pH 5.1)
- Detailed protocols (EnzChek Ultra Phytase Assay Kit)
Sufficient reagents are provided for five 96-well microplate assay with 6 mL per plate and 20% excess for pipetting considerations or partial-plate assays.
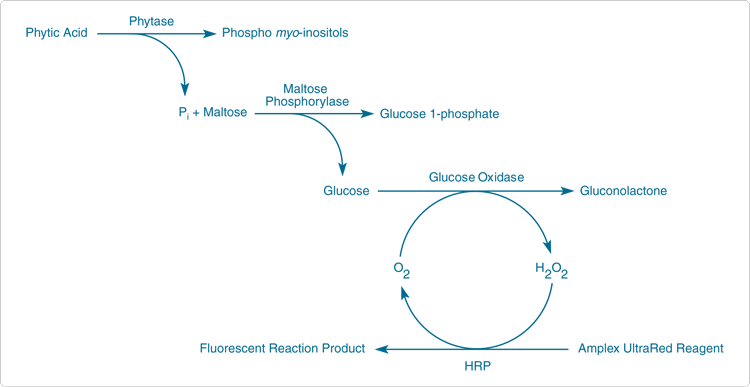
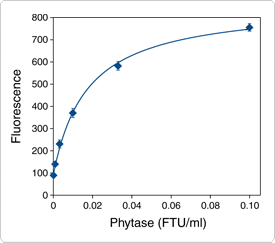
Figure 10.3.24 Assay of a standard phytase preparation using the EnzChek Ultra phytase assay. A dilution series of Natuphos 10000 L phytase (BASF) in 0.1 M acetate buffer (pH 5.5) was assayed with the Enzchek Ultra Phytase Assay Kit (E33701). Incubation time was 60 minutes; phytase activity is shown in FTU/mL (FTU= phytase unit, as defined by the manufacturer).
EnzChek Paraoxonase Assay Kit
Paraoxonase is a mammalian enzyme associated with high-density lipoprotein ("good cholesterol") in serum. Low serum paraoxonase levels are positively correlated with risk of cardiovascular disease,![]() and paraoxonase activity is a better marker than the PON1 genotype for predicting susceptibility to vascular disease.
and paraoxonase activity is a better marker than the PON1 genotype for predicting susceptibility to vascular disease.![]() Paraoxonase has multiple activities including organophosphatase, phosphotriesterase, arylesterase and thiolactonase. The organophosphatase activity confers protection against toxic organophosphates such as insecticides, which are a common source of chemical intolerance, as well as nerve agents such as sarin and VX. Current methods for measuring paraoxonase in serum, including the colorimetric paraoxon assay,
Paraoxonase has multiple activities including organophosphatase, phosphotriesterase, arylesterase and thiolactonase. The organophosphatase activity confers protection against toxic organophosphates such as insecticides, which are a common source of chemical intolerance, as well as nerve agents such as sarin and VX. Current methods for measuring paraoxonase in serum, including the colorimetric paraoxon assay,![]() are relatively insensitive and often toxic themselves, and are compromised by high background and low signal as well.
are relatively insensitive and often toxic themselves, and are compromised by high background and low signal as well.
The EnzChek Paraoxonase Assay Kit (E33702) is a highly sensitive, homogeneous fluorometric assay (excitation/emission maxima ~360/450 nm) for the organophosphatase activity of paraoxonase and is based on the hydrolysis of a fluorogenic organophosphate analog. Under standard conditions, this assay requires only 5 µL of serum, yields a signal in as little as 15 minutes and is linear for up to 60 minutes. The EnzChek paraoxonase assay is >10-fold more sensitive than the colorimetric paraoxon assay and, unlike the colorimetric assay, can distinguish samples of very similar paraoxonase activity. Furthermore, the EnzChek paraoxonase assay requires only a single homogeneous reaction, which may be either continuously monitored or terminated using a stop solution. Human serum organophosphatase activities measured using the EnzChek paraoxonase assay show correlation with paraoxonase concentrations determined by ELISA.![]() The Z' factor
The Z' factor ![]() (a statistical parameter for evaluating the signal window of an assay) is 0.95 when the assay is performed as described in the 96-well format.
(a statistical parameter for evaluating the signal window of an assay) is 0.95 when the assay is performed as described in the 96-well format.
Each EnzChek Paraoxonase Assay Kit provides:
- Paraoxonase substrate
- Fluorescent reference standard
- Concentrated reaction buffer
- Stop reagent
- 96-well microplates
- Adhesive microplate covers
- Organophosphatase positive control
- Dimethylsulfoxide (DMSO)
- Detailed protocols (EnzChek Paraoxonase Assay Kit)
Sufficient reagents are provided for 100 assays using a reaction volume of 110 µL per well and a fluorescence microplate reader.
The Antibody Beacon Tyrosine Kinase Assay Kit (A35725) provides a homogeneous solution assay for measuring the activity of tyrosine kinases and the effectiveness of potential inhibitors and modulators.![]() The key to this tyrosine kinase assay is a small-molecule tracer ligand labeled with our bright green-fluorescent Oregon Green 488 dye. When an anti-phosphotyrosine antibody binds this tracer ligand to form the Antibody Beacon detection complex, the fluorescence of the Oregon Green 488 dye is efficiently quenched. In the presence of a phosphotyrosine-containing peptide, however, this Antibody Beacon detection complex is rapidly disrupted, releasing the tracer ligand and relieving its antibody-induced quenching (Figure 10.3.25). Upon its displacement by a phosphotyrosine residue, the Oregon Green 488 dye–labeled tracer ligand exhibits an approximately fourfold fluorescence enhancement, enabling the detection of as little as 50 nM phosphotyrosine-containing peptide with excellent signal-to-background discrimination. Key benefits of the Antibody Beacon Tyrosine Kinase Assay Kit include:
The key to this tyrosine kinase assay is a small-molecule tracer ligand labeled with our bright green-fluorescent Oregon Green 488 dye. When an anti-phosphotyrosine antibody binds this tracer ligand to form the Antibody Beacon detection complex, the fluorescence of the Oregon Green 488 dye is efficiently quenched. In the presence of a phosphotyrosine-containing peptide, however, this Antibody Beacon detection complex is rapidly disrupted, releasing the tracer ligand and relieving its antibody-induced quenching (Figure 10.3.25). Upon its displacement by a phosphotyrosine residue, the Oregon Green 488 dye–labeled tracer ligand exhibits an approximately fourfold fluorescence enhancement, enabling the detection of as little as 50 nM phosphotyrosine-containing peptide with excellent signal-to-background discrimination. Key benefits of the Antibody Beacon Tyrosine Kinase Assay Kit include:
- Real-time measurements. Unlike many other commercially available tyrosine kinase assays, the Antibody Beacon Tyrosine Kinase Assay Kit permits real-time monitoring of kinase activity (Figure 10.3.26). Not only is the Antibody Beacon detection complex rapidly dissociated in the presence of phosphotyrosine residues, but the assay components have been designed to be simultaneously combined, eliminating any delay in the measurements.
- Simple detection protocol. Tyrosine kinase activity is measured by a simple increase in fluorescence intensity; no special equipment, additional reagents, or extra steps are required. This assay is readily compatible with any fluorescence microplate reader.
- Use of natural substrates. The Antibody Beacon tyrosine kinase assay utilizes unlabeled peptide or protein substrates, is compatible with substrates that are pre-phosphorylated at serine or threonine (but not at tyrosine) residues and is applicable to the assay of a wide variety of kinases.
- Compatibility. The anti-phosphotyrosine antibody provided in the Antibody Beacon Tyrosine Kinase Assay Kit is specific for phosphotyrosine residues; assay components such as ATP (up to 1 mM) and reducing agents such as dithiothreitol (DTT, up to 1 mM) do not interfere with this assay. This anti-phosphotyrosine antibody was selected from among several clones to produce the greatest fluorescence enhancement by the kinase-phosphorylated product.
- Reliability. This tyrosine kinase assay has a broad signal window, indicated by a Z' factor
of >0.85.
The Antibody Beacon Tyrosine Kinase Assay Kit comes with all the reagents needed to perform this assay, including:
- Oregon Green 488 dye–labeled tracer ligand
- Anti-phosphotyrosine antibody
- Concentrated tyrosine kinase reaction buffer
- Two generic tyrosine kinase substrate solutions: a poly(Glu:Tyr) solution and a poly(Glu:Ala:Tyr) solution
- Dithiothreitol (DTT)
- Adenosine triphosphate (ATP)
- Phosphotyrosine-containing peptide, phospho-pp60 c-src (521–533), for use as a reference
- Detailed protocols (Antibody Beacon Tyrosine Kinase Assay Kit)
Each kit provides sufficient reagents to perform ~400 assays using a 50 µL assay volume in a fluorescence microplate reader. We provide an extensive range of assays for protein kinases that utilize a variety of strategies to detect phosphorylation of peptide and protein substrates (Invitrogen kinase assay platforms—Table 17.1).
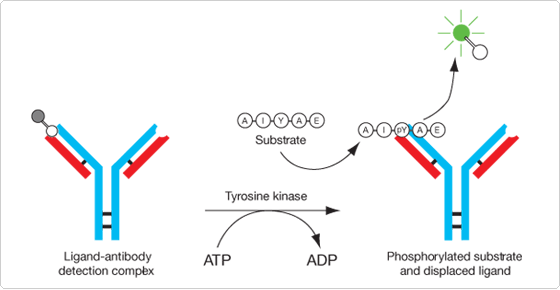
Figure 10.3.25 Reaction scheme for the tyrosine kinase assay used in the Antibody Beacon Tyrosine Kinase Assay Kit (A35725). The unlabeled natural substrate (AIYAE) is phosphorylated by the tyrosine kinase to AIY(P)AE, which displaces the quenched Oregon Green 488 dye–labeled peptide from the anti-phosphotyrosine antibody, resulting in a large increase in its fluorescence that is proportional to the amount of AIY(P)AE formed in the reaction.
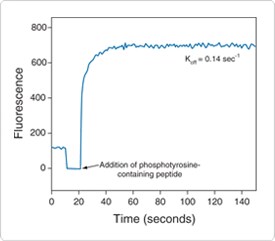
Figure 10.3.26 Real-time detection capability of the Antibody Beacon Tyrosine Kinase Assay Kit (A35725). Fluorescence of the Antibody Beacon detection complex in tyrosine kinase assay buffer was monitored over time. After ~15 seconds, an excess of phosphotyrosine-containing peptide was added to the Antibody Beacon detection complex and the off-rate was calculated
The ATP Determination Kit (A22066) is designed to provide a convenient method for the sensitive bioluminescence-based detection of ATP with recombinant firefly luciferase and its substrate luciferin. This assay is based on luciferase's absolute requirement for ATP to produce light. In the presence of Mg2+, luciferase catalyzes the reaction of luciferin, ATP and O2 to form oxyluciferin, AMP, CO2, pyrophosphate and ~560 nm light (Substrates for Microsomal Dealkylases, Acetyltransferases, Luciferases and Other Enzymes—Section 10.6).
The luciferin–luciferase bioluminescence assay is extremely sensitive; most luminometers can detect as little as 1 picomole of pre-existing ATP or ATP as it is generated in kinetic systems (Figure 10.3.27). This sensitivity has led to its widespread use for detecting ATP in various enzymatic reactions, as well as for measuring viable cell number ![]() (Viability and Cytotoxicity Assay Reagents—Section 15.2, Viability and Cytotoxicity Assay Kits for Diverse Cell Types—Section 15.3) and for detecting low-level bacterial contamination in samples such as blood, milk, urine, soil and sludge.
(Viability and Cytotoxicity Assay Reagents—Section 15.2, Viability and Cytotoxicity Assay Kits for Diverse Cell Types—Section 15.3) and for detecting low-level bacterial contamination in samples such as blood, milk, urine, soil and sludge.![]() The luciferin–luciferase bioluminescence assay has also been used to determine cell proliferation and cytotoxicity in both bacterial
The luciferin–luciferase bioluminescence assay has also been used to determine cell proliferation and cytotoxicity in both bacterial ![]() and mammalian cells,
and mammalian cells,![]() and to distinguish cytostatic versus cytocidal potential of anticancer drugs on malignant cell growth.
and to distinguish cytostatic versus cytocidal potential of anticancer drugs on malignant cell growth.![]()
Each ATP Determination Kit (A22066) contains:
- Luciferin (5 vials, each containing 3.0 mg)
- Luciferase
- Dithiothreitol (DTT)
- ATP
- Concentrated reaction buffer
- Detailed protocols for ATP quantitation (ATP Determination Kit)
Unlike most other commercially available ATP detection kits, our ATP Determination Kit provides the luciferase and luciferin packaged separately, which enables researchers to optimize the reaction conditions for their particular instruments and samples. The ATP Determination Kit provides sufficient reagents for performing 200 ATP assays using 0.5 mL sample volumes or 500 ATP assays using 0.2 mL sample volumes.
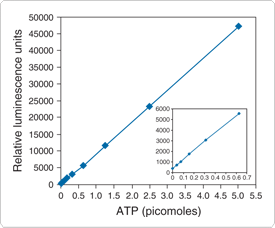
Figure 10.3.27 Detection of ATP using the ATP Determination Kit (A22066). Each reaction contained 1.25 µg/mL of firefly luciferase, 50 µM D-luciferin and 1 mM DTT in 1X reaction buffer. Luminescence was measured after a 15-minute incubation.
| Cat # | MW | Storage | Soluble | Abs | EC | Em | Solvent | Product | Notes |
|---|---|---|---|---|---|---|---|---|---|
| A22184 | 878.28 | FF,L | H2O | 504 | 68,000 | 514 | pH 7 | see Notes | 1, 2, 3 |
| D6487 | 422.20 | F,D,L | DMSO, H2O | 478 | 26,000 | 628 | pH 7 | DDAO (H6482) | |
| D6567 | 292.13 | F,D | DMSO, H2O | 320 | 14,000 | 385 | pH 9 | DiFMU (D6566) | 4 |
| D22065 | 292.13 | F,D | DMSO, H2O | 320 | 14,000 | 385 | pH 9 | DiFMU (D6566) | 4 |
| E6588 | 431.08 | F,L | H2O | 289 | 12,000 | see Notes | pH 10 | ELF 97 alcohol | 2, 4 |
| E6589 | 431.08 | F,L | H2O | 289 | 12,000 | see Notes | pH 10 | ELF 97 alcohol | 2, 4 |
| E6645 | 313.33 | FF,D | H2O | 332 | 16,000 | none | pH 7 | see Notes | 5, 6 |
| E6646 | 313.33 | F,D | H2O | 332 | 16,000 | none | pH 7 | see Notes | 5, 6 |
| F2999 | 560.39 | F,D | H2O | 272 | 5300 | none | MeOH | fluorescein (F1300) | 7 |
| G22183 | 894.28 | FF,L | H2O | 504 | 68,000 | 510 | pH 7 | see Notes | 1, 2, 3, 8 |
| M6491 | 256.15 | F,D | DMSO, H2O | 319 | 15,000 | 383 | pH 9 | HMC (H189) | 4 |
| N6495 | 817.65 | D,L | H2O, DMSO | 256 | 64,000 | none | MeOH | see Notes | 9 |
| |||||||||
For Research Use Only. Not for use in diagnostic procedures.
